What is the purpose of fixation for electron microscopy?
“..to preserve the structure of cells with minimum alteration from the living state with regard to volume, morphology, and spatial relationships of organelles and macromolecules, minimum loss of tissue constituents, and protection of specimens against subsequent treatments including rinsing, dehydration, staining, vacuum and exposure to the electron beam.” Hayat, 2000, Prinicpals and Techniques in Electron Microscopy. Biological Applications. 4th Edition. Cambridge University Press.
I don't think I can word the goals of fixation any better than outlined in the above quote. Ideally, fixation is as rapid as possible, and the techniques used need to be suited to the sample and instrumentation available. The above reference is an excellent resource on many techniques involved in preparing biological samples for electron microscopy and is highly recommended.
I don't think I can word the goals of fixation any better than outlined in the above quote. Ideally, fixation is as rapid as possible, and the techniques used need to be suited to the sample and instrumentation available. The above reference is an excellent resource on many techniques involved in preparing biological samples for electron microscopy and is highly recommended.
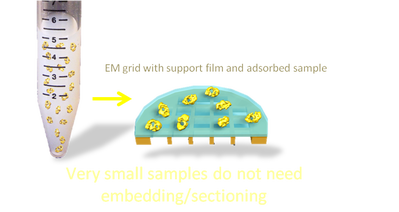
Viruses, proteins and samples less than ~300 nm in diameter.
Small samples do not require sectioning for TEM and can be adsorbed directly onto grids for TEM. The grids can be stained or frozen directly.
Chemical fixation of cells and tissues.
Cells can be grown on substrates such as glass cover slips or plastic discs designed for cell culture. The cells can be fixed directly on the substrate or detached and spun down into a pellet using a centrifuge.
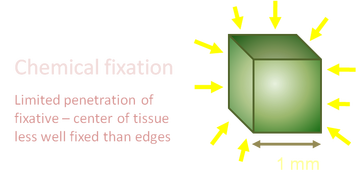
Tissue samples need to be dissected and cut into small pieces, approx 1-2 cubic mm before being immersed in the fixative solution. Chemicals used for fixation and staining can only penetrate tissue blocks and provide reasonable ultrastructural preservation to a depth of less than 2mm.
A technique called perfusion can be used to fix whole (anesthetised) animals via the circulatory system. The tissue is then dissected out and diced into small volumes before being placed into a fixative solution as in the immersion technique outlined above.
The length of chemical fixation is routinely two hours at room temperature. There are variations on this depending on temperature used (lower temperatures slow down the rate of fixative penetration, higher temperatures increase it). Microwaves can also be used to enhance the speed of fixation.
Tissue samples need to be dissected and cut into small pieces, approx 1-2 cubic mm before being immersed in the fixative solution. Chemicals used for fixation and staining can only penetrate tissue blocks and provide reasonable ultrastructural preservation to a depth of less than 2mm.
A technique called perfusion can be used to fix whole (anesthetised) animals via the circulatory system. The tissue is then dissected out and diced into small volumes before being placed into a fixative solution as in the immersion technique outlined above.
The length of chemical fixation is routinely two hours at room temperature. There are variations on this depending on temperature used (lower temperatures slow down the rate of fixative penetration, higher temperatures increase it). Microwaves can also be used to enhance the speed of fixation.
- Chemical cross-links are formed between molecules
- Chemical fixation provides long term stability and electron contrast
- It is widely available for little cost
- Most protocols are well established for a range of samples/questions
- It causes artefacts
- The chemicals used are hazardous to human health
Cryo-fixation
Freezing biological samples is an excellent way to halt cellular processes as fast as possible. It is a technically challenging technique, however, and requires equipment that is often expensive.
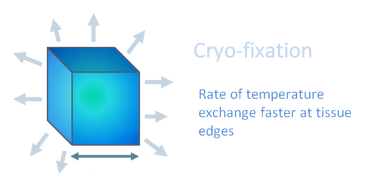
As with chemical fixation, it is important that the sample size is minimised. Samples are frozen by a process of rapid cooling, the rate of which will be fastest at the edge of the sample. The aim of cryo fixation is to VITRIFY the water in the sample, changing the state of the water from a liquid to a solid with no intermediate ice-crystal formation. When the rate of cooling is not fast enough, ice-crystal formation causes a segregation between pure water and solute, creating an artifactual compartmentalisation of cellular components. Ice-crystals also disrupt the organelles and membranes within a cell, changing its structure.
There are several freezing techniques that provide rapid cooling of the sample.
PLUNGE FREEZING - small samples (those adsorbed onto grids as described above) and plunged into propane or ethane that is previously cooled by and is sitting in a bath of liquid nitrogen.
"SLAM" FREEZING - samples are rapidly brought into contact with a metal block that has been cooled using liquid nitrogen.
HIGH PRESSURE FREEZING - a high pressure (approx. 2000 bar) is applied to the sample immediately prior to freezing. This changes the physical properties of water enough to reduce the range of temperatures over which ice-crystals may occur.
As with chemical fixation, it is important that the sample size is minimised. Samples are frozen by a process of rapid cooling, the rate of which will be fastest at the edge of the sample. The aim of cryo fixation is to VITRIFY the water in the sample, changing the state of the water from a liquid to a solid with no intermediate ice-crystal formation. When the rate of cooling is not fast enough, ice-crystal formation causes a segregation between pure water and solute, creating an artifactual compartmentalisation of cellular components. Ice-crystals also disrupt the organelles and membranes within a cell, changing its structure.
There are several freezing techniques that provide rapid cooling of the sample.
PLUNGE FREEZING - small samples (those adsorbed onto grids as described above) and plunged into propane or ethane that is previously cooled by and is sitting in a bath of liquid nitrogen.
"SLAM" FREEZING - samples are rapidly brought into contact with a metal block that has been cooled using liquid nitrogen.
HIGH PRESSURE FREEZING - a high pressure (approx. 2000 bar) is applied to the sample immediately prior to freezing. This changes the physical properties of water enough to reduce the range of temperatures over which ice-crystals may occur.
- Rapid freezing to achieve vitrification
- Tissue can be imaged in an hydrated state in microscopes with cryo stages
- Expensive equipment required and might not be available
- Technically challenging methods take time to master
- Not suited to all sample types